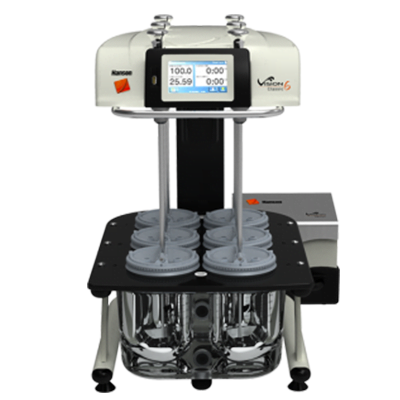
The Teledyne Hanson 14-vessel CD14 comparative dissolution tester runs two methods simultaneously or independently. It’s ideal for bioequivalence studies of generic vs. innovator drug formulations.
With 14 stirring positions, the CD14 supports 6+1 or 12+2 configurations, improving workload efficiency in R&D, formulations, QC, and stability-test labs.
The CD14 is controlled by a fast, powerful computer built into the color touchscreen display mounted to the drive head. Its adjustable tilt angle accommodates operators of various heights. The Linux-based operating system, equipped with its own SQL database responds instantly to user input. The highly durable resistive touchscreen works with gloves on or off.
An easy-to-use interface includes context-sensitive help; logs for events, errors, test reports, test history, and service; rapid programming of new methods; ability to search the database by any combination of fields; import/export including full system backup; programmable alarms for automatic power-on and warm-up; countdown to sampling points; tools including full system diagnostics; and more.
Features:
- The CD14 provides the smallest footprint in the 14-vessel class: 85 cm W x 61 cm D.
- Precision vessel centering system exceeds the most demanding enhanced mechanical specifications.
- Store up to 500 protocols (methods) on-board.
- Run two different 6-vessel methods simultaneously or one 12-vessel method.
- Rugged waterbath with low-position front port for ease of draining.
- Heater/circulator slides out for ease of servicing.
- Large color touchscreen with adjustable tilt angle for operators of different heights.
- Compatible with Apparatus 1, 2, 5, and 6.
- Spindle speed: 25 rpm to 250 rpm, accurate to ± 1 rpm with displayed resolution 0.1 rpm.
- Temperature: programmable from 25.0 °C to 50.0 °C with accuracy of ± 0.5 °C.
- Digital temperature probes: 10 °C to 60 °C with accuracy of ± 0.1 °C from 30 °C to 55 °C, and displayed resolution of 0.01 °C.
- Compliant with USP, EP, JP, US FDA Enhanced Mechanical Calibration and ASTM E2503 standards.
- Seamless integration with CD AutoPlus™ autosamplers and AutoFill™ fraction collectors.
21 CFR PART 11 compliance
The CD14 onboard database stores up to 100 users with three levels of security (operators, managers, and administrators) with the ability to customize permissions and profiles at each level. The CD14 biometric fingerprint reader allows rapid enrolment of one or two fingers and provides positive ID of instrument operators. A password-only option is also included. Customize your own report formats and approval systems by specifying up to three signatures per report. An extensive audit trail records all system events and logs any changes made by users. System configurations are stored on-board and the system logs any changes to methods and configurations.
Fast, Powerful Computer Control
The CD14 is controlled by a fast, powerful computer built into the color touch screen display mounted to the drive head. The adjustable tilt angle accommodates operators of different heights while the Linux-based operating system with its own SQL database responds instantly to user input. The highly durable resistive touch screen works with gloves on or off. An easy-to-use interface includes context-sensitive help; logs for events, errors, test reports, test history, and service; rapid programming of new methods; ability to search database by any combination of fields; import/export including full system backup; programmable alarms for automatic power-on and warm-up; countdown to sampling points; tools including full system diagnostics; and more.
Find the perfect Dissolution Testing Solution for your Needs

With this user-friendly tool, you can effortlessly design a system that perfectly aligns with your specific needs, ensuring maximum efficiency and productivity. From selecting dissolution baths to choosing specific accessories, the possibilities are endless!